|
There are some systematic deviations from this trend, however. Down a group, the IE 1 value generally decreases with increasing Z. Within a period, the IE 1 generally increases with increasing Z. The values of first ionization energy for the elements are given in Figure 5. Relating this logic to what we have just learned about radii, we would expect first ionization energies to decrease down a group and to increase across a period.įigure 4 graphs the relationship between the first ionization energy and the atomic number of several elements. Thus, as size (atomic radius) increases, the ionization energy should decrease. For larger atoms, the most loosely bound electron is located farther from the nucleus and so is easier to remove. Energy is always required to remove electrons from atoms or ions, so ionization processes are endothermic and IE values are always positive. The energy required to remove the third electron is the third ionization energy, and so on. For all other atoms, the inner electrons partially shield the outer electrons from the pull of the nucleus, and thus: For hydrogen, there is only one electron and so the nuclear charge ( Z) and the effective nuclear charge ( Z eff) are equal. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. This can be explained with the concept of effective nuclear charge, Z eff. This might seem counterintuitive because it implies that atoms with more electrons have a smaller atomic radius. Within each group (e.g., the alkali metals shown in purple), the trend is that atomic radius increases as Z increases.Īs shown in Figure 2, as we move across a period from left to right, we generally find that each element has a smaller covalent radius than the element preceding it. Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr. Covalent Radii of the Halogen Group Elementsįigure 2. The trends for the entire periodic table can be seen in Figure 1. This trend is illustrated for the covalent radii of the halogens in Table 1 and Figure 1. Consequently, the size of the atom (and its covalent radius) must increase as we increase the distance of the outermost electrons from the nucleus. Thus, the electrons are being added to a region of space that is increasingly distant from the nucleus. We know that as we scan down a group, the principal quantum number, n, increases by one for each element. We will use the covalent radius (Figure 1), which is defined as one-half the distance between the nuclei of two identical atoms when they are joined by a covalent bond (this measurement is possible because atoms within molecules still retain much of their atomic identity). However, there are several practical ways to define the radius of atoms and, thus, to determine their relative sizes that give roughly similar values. The quantum mechanical picture makes it difficult to establish a definite size of an atom. With just a few clicks, you can create three-dimensional versions of the periodic table showing atomic size or graphs of ionization energies from all measured elements. They are (1) size (radius) of atoms and ions, (2) ionization energies, and (3) electron affinities.Įxplore visualizations of the periodic trends discussed in this section (and many more trends) on the Atomic Number of the Elements website. 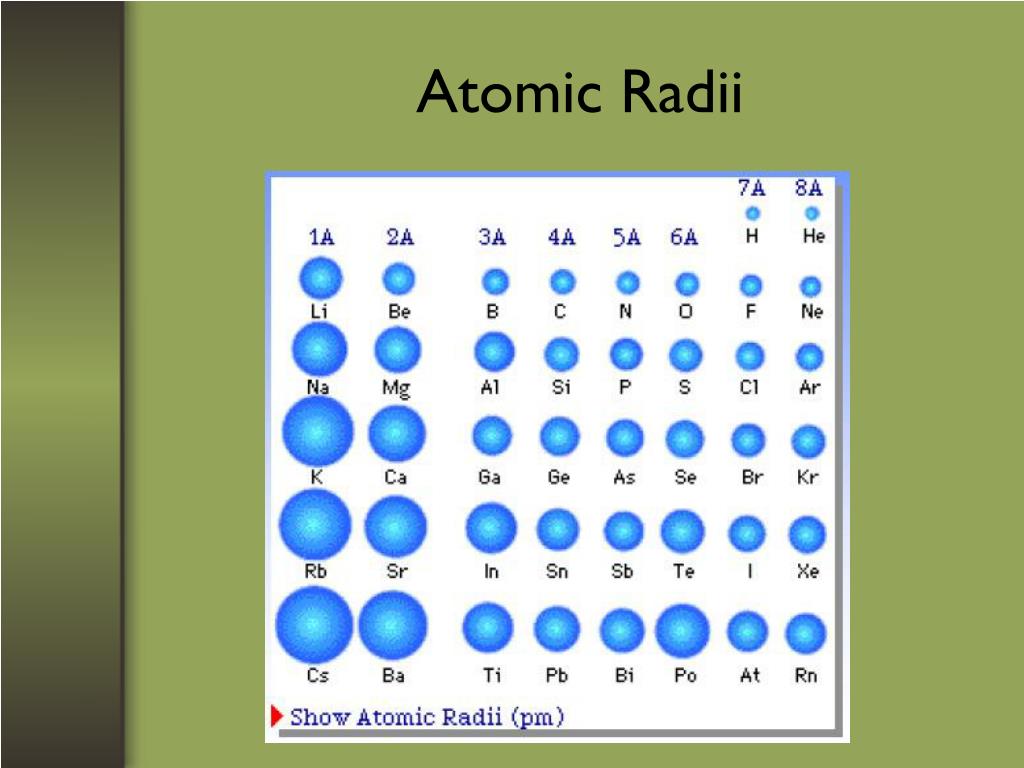
These properties vary periodically as the electronic structure of the elements changes. An understanding of the electronic structure of the elements allows us to examine some of the properties that govern their chemical behavior. As we go down the elements in a group, the number of electrons in the valence shell remains constant, but the principal quantum number increases by one each time. Oxygen, at the top of group 16 (6A), is a colorless gas in the middle of the group, selenium is a semiconducting solid and, toward the bottom, polonium is a silver-grey solid that conducts electricity.Īs we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. For example, as we move down a group, the metallic character of the atoms increases. 
However, there are also other patterns in chemical properties on the periodic table. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
